Growing Cannabis in Hard water
Growing Cannabis in Hard water and what you can do about it. Eighty-five percent of the United States has water that is hard sufficient to need softening. The chemistry of difficult water is not complicated, it is merely an abundance of calcium (Ca) and magnesium (Mg)– typically called “hardness minerals”– dissolved in water.

In contrast rain is naturally soft water however it does not remain soft for long; as the rain falls on the earth it permeates through soil and rock which is filled with solidity minerals. The rainwater soon turns from soft to hard. The level of firmness depends upon the makeup of the rock in your water catchment area. All rock minerals have an electrical charge, both the solidity minerals Ca and Mg are positively charged, this a fundamental part in understanding how a water conditioner works.
But what is hard water?
Hard water has a high concentration of dissolved mineral salts, especially calcium and magnesium. Hardness is measured in milligrams of calcium carbonate per liter (mg / L); ppm in the US (parts per million). Depending on how high the concentration of calcium and magnesium, a distinction is made between the following types of water:
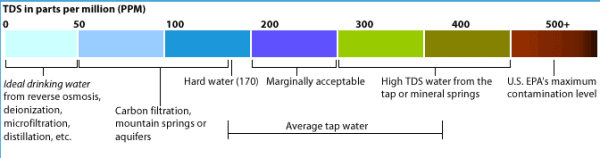
- soft water: has a concentration of less than 150 mg / L calcium carbonate
- semi-soft water: has a concentration between 150-200 mg / L calcium carbonate
- hard water: has a concentration between 200-400 mg / L calcium carbonate
- very hard water: has a concentration between 400-550 mg / L calcium carbonate
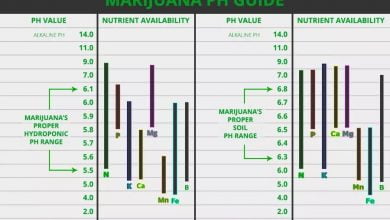
Hard water can cause problems in the cultivation of cannabis. Calcium deposits in the roots cause nutrients from the plant can no longer be absorbed correctly. It may happen that the nutrient given then have little or no desired effects. If fertilizers are given with additional calcium and magnesium in combination with hard water, it may even lead to nutrient blockage in the roots of plants. All nutrients are then locked outright and no longer allowed in the plant. The respective dissolved salt content of the water depends on where you and your plants live. By nature, the plants are watered with rainwater, which is soft.
Factors that affect water quality may be: living near mountains, by the sea or in major cities. Whenever the water is too hard, it should be softened for cannabis plants. They are among the plants that can not deal with hard water. In most European regions and cities hard water is more of a problem. Few regions can look forward to soft water from the tap.
An excessive amount of calcium and magnesium in the water can thus cause nutrient defects in cannabis plants. The only reliable method to remove dissolved salts to be removed from the water, the Reverse Osmosis System .
Why use a Reverse Osmosis System in cannabis cultivation and what are the benefits?
- By using a reverse osmosis plants Cannabis plants are healthier and get a stronger root system
- nutrients can be taken up to 100%, thus there is an increase in income
- mineral salt deposits in the roots are avoided
- nozzles and hoses clog less in hydroponic systems and have a longer life
In some countries, the water quality is very high and tap water can be drunk without any health concernes. A high calcium or magnesium content, which may also be present in the water is not in any way a concern for humans. Humans have a very complex digestive system that can cope well with these amounts of minerals and are actually required. Cannabis plant are a little different, they are more sensitive to excessive amounts of certain minerals Briefly: bottled water or tap water of drinking water quality is suitable for humans, but not always for cannabis plant
How does reverse osmosis work?
Osmosis is the concentration balance of two liquids through a semipermeable membrane. That always happens when two aqueous liquids with different ion concentration are separated by a semi-permeable wall. In nature, this principle plays an important role. A semi-permeable membrane only the solvent but not the solids are allowed through.
In this way, a regulation of water of the cells is possible, and will also receive the internal pressure. Ion solutions, which are separated by a membrane, always want to achieve a concentration equalization. The membrane is a barrier to ions with too high a molecular size. This ion membrane is not easily penetrate. So the smaller water molecules from the low concentrated side migrate to the more concentrated side.
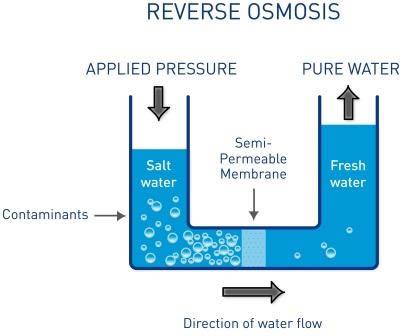 The water molecules move as long until either the ion concentration on both sides is equalized or a pressure on the high concentration side is applied. This is called osmotic pressure. In reverse osmosis, this principle is reversed. On the side of the high ion concentrations (tap water), a pressure is generated which forces the water towards the pure water to the other side. Unwanted salts such as hardness components (calcium, magnesium) or harmful substances such as pesticide residues or contaminants, because of their molecular size cannot pass through the extremely fine membrane.
The water molecules move as long until either the ion concentration on both sides is equalized or a pressure on the high concentration side is applied. This is called osmotic pressure. In reverse osmosis, this principle is reversed. On the side of the high ion concentrations (tap water), a pressure is generated which forces the water towards the pure water to the other side. Unwanted salts such as hardness components (calcium, magnesium) or harmful substances such as pesticide residues or contaminants, because of their molecular size cannot pass through the extremely fine membrane.
The trapped materials from the membrane must be removed continuously, so that the membrane does not become clogged.
- reverse osmosis water (demineralized water) has a pH of 7 GPG (grain per gallon), but only for a short time. Demineralized water is slightly acidic (4,5 – 5,0).

.
Important tips for using a reverse osmosis system
Calcium happens throughout the whole plant. It is utilized for lots of procedures in the plant, however, calcium is most important for the development process. It has a managing impact in the cells and contributes to the stability of the plant. Plants have two transport systems at their disposal: the xylem vessels and the sieve vessels. The majority of nutrients can be carried via both systems, nevertheless, for calcium this is not possible.
If you are using a reverse osmosis system to produce water for your plants, it may be necessary to use a magnesium and calcium supplements: for example, so that no deficiencies arise. In general, manufacturers expect growers to be using tap water, that already has calcium and magnesium (a calcium and magnesium supplement would be important if osmosis water is used). When manufacturers devise fertilizing programs for hydroponic cultivation it is often assumed 000ppm water, meaning that the nutrients are formulated for pure water (In this case extra calcium or magnesium would then in most cases be not necessary).
The filters of reverse osmosis units should be regularly cleaned and replaced in order to ensure their full functionality. This differs from one manufacturer to another and also depends on their use, normally the filters are replaced every 4-6 months.
reverse osmosis water must not be used in pure form. It can be 1: 1 or 2: 1 mixed with normal tap water to obtain ideal watering for the plants. The mixing ratio depends on how much calcium and magnesium is contained in the supply water. When using pure water (000ppm) as described above, magnesium or calcium should be added to prevent deficiencies.